 |
 |
 |
September 4, 2006
| Toray Industries, Inc. |
| Japan Tobacco Inc. |
| Torii Pharmaceutical Co., Ltd. |
|
 |
Toray, JT, Torii Agree to Co-Develop and Market
TRK-820 Antipruritus Drug for Hepatic Disease Patients in Japan |
 |
September 4, 2006 - Tokyo, Japan: Toray Industries, Inc. ("Toray") [TSE: 3402], Japan Tobacco Inc. ("JT") [TSE: 2914] and Torii Pharmaceutical Co., Ltd. ("Torii") [TSE: 4551] announced today a new agreement to jointly develop and market a novel antipruritic agent, TRK-820, discovered at Toray's Pharmaceutical Research Laboratories for hepatic disease patients in Japan. This is in addition to the existing co-development arrangement with the same parties on the same compound for a treatment of uremic pruritus for hemodialysis patients.
Hepatic disease-related pruritus is a non-inflammatory systemic or local severe itching condition with an unclear pathology. The patients with serious itching condition experience sleep disturbances, which subsequently causes various quality of life (QOL) problems. It has been known that conventional antipruritic agents are not fully effective in treating this condition, and an effective treatment has long been awaited.
TRK-820 is a selective 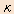 (kappa) opioid receptor agonist with a novel mechanism of action which differs from that for conventional drugs such as antihistamine, and it is expected that the compound will be effective for treating itching that is resistant to existing therapeutics. (kappa) opioid receptor agonist with a novel mechanism of action which differs from that for conventional drugs such as antihistamine, and it is expected that the compound will be effective for treating itching that is resistant to existing therapeutics.
With respect to the new agreement, TRK-820 will be co-developed by Toray and JT, and marketed by Torii in the area of hepatic disease-related pruritus as well as uremic pruritus in hemodialysis. The on-going clinical development of TRK-820 for uremic pruritus in hemodialysis patients by collaboration of these three companies is in the final stage of the pivotal study, which is being advanced as scheduled.
The following notes provide additional information about TRK-820.
Notes:
|
|
| 1. |
Characteristics of TRK-820 |
TRK-820 is a novel selective 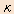 (kappa) opioid receptor agonist, with a different mechanism from existing antipruritic drugs such as antihistamine. The drug is thought to suppress the pruritus mediated by the opioid-related mechanism and hence is expected to be effective in treating itching that is resistant to existing treatments such as antihistamine drugs. (kappa) opioid receptor agonist, with a different mechanism from existing antipruritic drugs such as antihistamine. The drug is thought to suppress the pruritus mediated by the opioid-related mechanism and hence is expected to be effective in treating itching that is resistant to existing treatments such as antihistamine drugs. |
 |
| 2. |
Opioid receptors |
Receptors are the molecules located on the cell membranes. When receptors are bound by the medical compounds, a biological response is triggered. There are a wide variety of receptors in the human body and each of them has a different specificity to its binding compounds and pharmacological functions expressed by such bindings. An opiate is one such medical compound that binds to a receptor and expresses a response, and its receptors are known as opioid receptors. Whereas it was thought that there was only one type of opioid receptor when it was initially discovered, the subsequent evolution of the research in the area revealed that it is largely classified into three receptor types indicated as 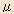 (mu), (mu), 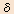 (delta) and (delta) and 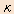 (kappa). (kappa). |
 |
| 3. |
Agonists |
| Pharmacological molecules which target receptors are divided into agonists and antagonists. An agonist activates or stimulates, and thereby induces either an increase or decrease in the biological activities by the cell. |
 |
| Corporate Profiles |
|
| Toray Industries, Inc. |
 |
Headquarters: |
1-1, Nihonbashi-Muromachi 2-chome, Chuo-ku, Tokyo |
 |
President: |
Sadayuki Sakakibara |
 |
Capital: |
96.937 billion yen |
 |
| Japan Tobacco Inc. |
 |
Headquarters: |
2-1, Toranomon 2-chome, Minato-ku, Tokyo |
 |
President: |
Hiroshi Kimura |
 |
Capital: |
100 billion yen |
 |
| Torii Pharmaceutical Co., Ltd. |
 |
Headquarters: |
Torii Nihonbashi Bldg., 4-1, Nihonbashi-Honcho 3-chome, Chuo-ku, Tokyo |
 |
President: |
Norihiko Matsuo |
 |
Capital: |
5.19 billion yen |
 |
|
|
 |
| |
|
|
 |
 |